December 2014 VOLUME 2/2014 PHARMACY BULLETINhpermai.moh.gov.my/sites/default/files/docs/Bulletin...
Transcript of December 2014 VOLUME 2/2014 PHARMACY BULLETINhpermai.moh.gov.my/sites/default/files/docs/Bulletin...

PHARMACY BULLETIN VOL. 2/2014 DECEMBER 2014
Highlight Issue: Ebola
Medication Safety: Look Alike Medications
Adverse Drug Reaction: Methylphenidate
Your Medicine: Asenapine
Service in Pharmacy: Pharmacy Appointment Card
Counselling: Nicotine Patch
Pharmacy Event: Aktiviti– Aktiviti Kenali Ubat Anda 2014
Drug in Hospital Permai Johor Bahru: New drug in the formulary
Fun Corner: Pharmacy Word Search
December 2014 VOLUME 2/2014
PHARMACY BULLETIN
E D I T O R I A L B O A R D A D V I S O R :
N o o r R a t n a N a h a r u d d i n
C H I E F E D I T O R :
H a f i z a h b i n t i H a m i d i
N o o r a n i S a l i m a h b i n t i S a k h o n
C O - E D I T O R S :
C h u a Y e e M i n
F o o Q i n H a o
G a n S u L i n g
A b d u l H a k i m N o r d i n
N u r S y a f i q a h M a ’ o n
T P O S I C

PHARMACY BULLETIN VOL. 2/2014 DECEMBER 2014
EBOLA
Background of Ebola virus Ebola virus was first found in 1976 in Zaire (now known as the Democratic Republic of Congo). However,
the name of the virus was then changed to Ebola in 2010 which signifies a river there. There are 5 Ebola
virus subtypes namely: Ebola-Zaire, Ebola-Sudan, Ebola-Ivory Coast, Ebola-Bundibugyo and Ebola-Reston.
Out of these virus subtypes, all except Ebola-Reston is dangerous to human.
Spread of Ebola Virus The first outbreak of Ebola started in West Africa in 4 countries (Guinea, Sierra Leone,
Liberia and Nigeria). The likely host are bats. Ebola virus disease only spreads when
people are sick so a patient must have the symptoms mentioned above to spread the
disease to others. After 21 days, if an exposed person does not develop symptoms,
they will not become sick with Ebola.
Means of spread of Ebola virus:
Contact with bodily fluid of a person who is sick with or has died from Ebola (blood, vomit, faeces,
urine, sweat, semen, spit or other body fluids).
Objects contaminated with the virus (needles, medical equipment)
Infected animals (by contact with blood or fluids or infected meat).
Ebola in Malaysia Malaysians were shocked with the news of a suspected Ebola virus disease case in Kuching, Sarawak in September 2014 but the Chief Director of Ministry of Health, Datuk Dr Noor Hisham Abdullah have confirmed that after close investigation, the patient is clear of Ebola virus.
Symptoms of Ebola virus If any of these symptoms develop,
seek immediate medical attention and
inform of previous exposures such as
previous visit to the affected regions
such as West Africa, close contact with
an infected person, handled the
remains of someone affected or
recently travelled in the same carrier as
someone who is suspected or infected
with Ebola virus
Prepared By : Chua Yee Min Page 1
Highl i gh t I s sue :

PHARMACY BULLETIN VOL. 2/2014 DECEMBER 2014
Patient Management Supportive care: the most important aspect of supportive care
involves preventing intravascular volume depletion, correcting
profound electrolyte abnormalities and avoiding the
complications of shock.
Careful haemodynamic monitoring needed and intravenous
fluid depletion requirements may be high (5 to 10 liters per
day). Ebola virus disease may result in reduced effective
arterial blood volume despite extracellular fluid volume
overload (third spacing)
Patients may develop significant electrolyte disturbances
(eg, hyponatremia, hypokalemia) and may require frequent
repletion of electrolytes to prevent cardiac arrhythmias.
Patients may require nutritional support.
Isolation room (single patient room containing a private
bathroom) with the door closed. Facilities should maintain a
log of everyone entering the patient’s room.
Intensive nursing may be required in order to respond to patient’s changing clinical situation.
Additional measures: correction of severe coagulopathy and symptomatic management of fever,
nausea, vomiting, diarrhea and abdominal pain.
Empiric antimicrobial treatment should be considered
when patients develop vomiting, diarrhea and/or other
signs of severe gastrointestinal dysfunction and/or signs
of sepsis.
Prevention Always wash your hands with soap and rinse thoroughly
with water
Always cook your food properly.
If any of the symptoms of EVD develop, seek immediate
medical attention.
Do not touch people with signs of Ebola or have died from
Ebola.
Do not touch clothes and beddings of people who have
died from Ebola.
Do not touch the bodily fluids of people who have symp-
toms of Ebola.
Do not play with monkeys and baboons.
Do not eat fruits that have been eaten by bats.
References:
Centre of Disease Control and Prevention (CDC) United States of America on Ebola, retrieved 11 November 2014.
World Health Organisation (WHO) on Ebola, retrieved 11 November 2014.
UpToDate 2014 on Ebola and management of Ebola, retrieved 11 November 2014.
Ministry of Health Malaysia, retrieved 11 November 2014 (http://kpkesihatan.com/category/communicable-disease/ebola/)
Prepared By : Chua Yee Min Page 2

PHARMACY BULLETIN VOL. 2/2014 DECEMBER 2014
Look Alike medications
Left: T. Folic Acid 5mg
Right: T. Vitamin B complex
Left: T. Paracetamol 500mg
Right: T. Lithium Carbonate
300mg
Up: T. Clozapine 25mg Down: T. Chlorpheniramine 4mg
Up: T. Hyoscine Butylbromide 10mg
Down: T. Chlorpromazine 25mg
Left: T. Olanzapine Zydis 5mg Right: T. Olanzapine Zydis 10mg
Left: T. Prolase
Right: T. Dothiepin 25mg
Up: T. Albendazole 200mg
Down: T. Co-Trimoxazole
Up: T. Indomethacin 25mg Down: T. Atomoxetine 25mg
Left: T. Atenolol 100mg Right: T. Ascorbic Acid 100mg
Up: T. Fluoxetine 20mg Down: T. Haloperidol 5mg
Prepared by : Foo Qin Hao Page 3
Medication Safety:

PHARMACY BULLETIN VOL. 2/2014 DECEMBER 2014
The U.S. Food and Drug Administration (FDA) is warning that methylphenidate products, one type of central nervous system stimulant drug used to treat attention deficit hyperactivity disorder (ADHD), may
in rare cases cause prolonged and sometimes painful erections known as priapism. Priapism is a
condition where when blood in the penis becomes trapped, leading to an abnormally long-lasting and sometimes painful erection. This can occur in male patients of any age. Patients who take methylphenidate and develop erections lasting longer than four hours should seek immediate medical treatment. If not treated right away, priapism can lead to permanent damage to the penis. Review of post marketing reports by FDA showed that priapism usually developed some time after patients started tak-ing the drug, after an increase in dose, or during a period of drug withdrawal.
Safety Alert
Currently in Malaysia, there are 12 registered products containing methylphenidate. It is listed in the Ministry of Health Drug Formulary under category A (to be prescribed by consultants or specialists only).
The Drug Safety Monitoring Centre, National Pharmaceutical Control Bureau (NPCB) has received a total of 32 ADR reports related to methylphenidate with 48 adverse events. There were no reports of priapism or related events in Malaysia. Adverse events reported included nausea, vomiting, rash and other skin reactions, increase in liver enzymes and psychiatric disorders such as agitation. Direct Healthcare Professional Communications (DHPCs) have been approved by the NPCB in April &July 2014 to inform healthcare professionals regarding this safety issue. A directive has also been issued for the package inserts of all products containing methylphenidate to be updated with in-formation on the risk of priapism.
Background
Local Setting Advice to healthcare
providers
There is a rare risk of priapism associated with the use of methylphenidate.
Patients using methylphenidate should not stop taking it without first talking to their healthcare professionals.
All male patients and their caregivers should be counselled on the signs and symptoms of priapism and the importance of seeking immediate medical treatment if this occurs.
All ADRs suspected to be related to the use of methylphenidate use should be reported to the Drug Safety Monitoring Centre, NPCB.
Prepared by: Foo Qin Hao Page 4
Methylphenidate Adverse Drug Reaction:

PHARMACY BULLETIN VOL. 2/2014 DECEMBER 2014
Asenapine (Saphris®) is a new atypical
antipsychotic indicated for manic/ mixed
episodes associated with bipolar I disorder and
schizophrenia. Asenapine is available in 5 and
10mg fast-dissolving sublingual tablets
ASENAPINE — A NOVEL ANTIPSYCHOTIC FOR
SCHIZOPHRENIA AND MIXED OR MANIC EPISODES OF BIPOLAR
Introduction
Pharmacokinetics
Bioavailability 35% (Sublingual),
reduced to 2% (if
swallowing)
Time to onset of
action
3 minutes
Time to peak plasma
concentration
30-90 minutes
Half life 24 hour
Time to steady state 3 days
Dosage
Indication Initial dose Maintenance dose
Schizophrenia 5mg BD 10 mg BD
Bipolar 10mg BD (5mg BD if
use with lithium/
valproate)
10 mg BD
Dosage adjustment
renal or mild -moderate hepatic impairment Not required
severe hepatic impairment Avoid Asenapine
adverse effect not tolerated reduced to 5mg twice daily
Elderly (dementia) Contraindicated
Prepared by: Gan Su Ling Page 5
Adverse Effects
The commonly reported adverse effects are somnolence, dizziness, extrapyramidal side effects,
restlessness, oral hypoesthesia, weight gain, elevated prolactin and QT prolongation. In a 52 weeks
double-blind study by Shoemaker et al, the metabolic effect of Asenapine was studied where the
differences from baseline in total cholesterol and glucose levels were found to be not significant. The
study also showed that the proportion of other side effects is lower in patients taking Asenapine as
compared to other atypical antipsychotic such as olanzapine and risperidone.
Your Medicine:

PHARMACY BULLETIN VOL. 2/2014 DECEMBER 2014
Drug Interactions
Drugs Description
CYP1A2 Inhibitor Fluvoxamine
Imipramine
Increase asenapine concentration
QT prolongation agents
Amiodarone Quinidine
Chlorpromazine Ziprasidone
Avoid use in combination
In conclusion, Asenapine is a new atypical antipsychotic which could be advantageous, among
non-compliant or acutely agitated patients, due to its readily-dissolved and rapidly-absorbed sublingual
formulation. Asenapine also poses treatment advantage in patients who are unable to tolerate the
metabolic side effects which are common concerns for majority antipsychotics.
References
Cetin M. Asenapine: A novel hope in the treatment of schizophrenia and manic and mixed episodes of bipolar I disorder. Bulletin of Clinical Psychopharmacology. 2013. 23(1): 99-106.
Drug Information Handbook. 22th ed. Hudson, Ohio, Lexi-Comp, Inc.;2013:17
Prepared by: Gan Su Ling Page 6
Counselling Points

PHARMACY BULLETIN VOL. 2/2014 DECEMBER 2014
Prepared by: NurSyafiqah Ma’on Page 7
Bahagian Perkhidmatan Farmasi telah
menawarkan Perkhidmatan Tambah Nilai melalui
Sistem Temujanji Farmasi (STF) di hospital-hospital
kerajaan dan klinik-klinik kesihatan selain daripada
Sistem Pendispensan Ubat Bersepadu (SPUB),
SMS & Ambil, Pandulaju dan Ubat Melalui Pos
(UMP) bagi meningkatkan penggunaan ubat-ubatan
secara berkualiti dalam kalangan rakyat Malaysia.
Perkhidmatan ini membolehkan pesakit mengambil
bekalan ubat susulan di kaunter farmasi pesakit
luar tanpa perlu menunggu lama kerana ubat
pesakit akan disediakan terlebih dahulu sebelum
tarikh kedatangan pesakit. Pesakit akan dibekalkan
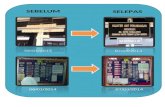
dengan sekeping kad temujanji (rujuk Gambar 1)
sebagai rujukan tarikh pengambilan ubat di kaunter
farmasi. Perkhidmatan ini disediakan bagi pesakit
kronik yang stabil, yang telah dibekalkan dengan
tempoh preskripsi melebihi sebulan.
Walaubagaimanapun, pembekalan ubat untuk bu-
lan pertama masih lagi perlu dilakukan seperti biasa
di kaunter farmasi pesakit luar.
Di Hopital Permai Johor Bahru, kad
temujanji banyak digunakan bagi pesakit-pesakit
yang datang dari rumah jagaan, rumah kebajikan,
atau penjara atau pesakit dari Clozapine Klinik.
Sistem Kad Temujanji
Farmasi Pesakit Luar
Contoh Kad Temujanji Farmasi Pesakit Luar
Beri penerangan tentang Kad Temu-janji selepas pendispensan ubat
Maklumkan kepda pe-sakit tarikh pengambi-lan ubat akan datang Pesakit setuju?
Isi keterangan pesakit pada Kad
Maklumkan kepada pesakit tarikh
Serahkan Kad Temujanji dan pris-kripsi asal kepada pesakit
Simpan salinan preskripsi ke dalam ‘Fail Kad Temujanji’ dan rekod
maklumat pesakit dalam ‘Buku Rekod
Tidak
Ya
Terima Kad Temujanji di kaunter
Cari ubat pesakit yang telah dise-diakan mengikut no. rujukan Kad
Temujanji
Pembekalan penuh?
Simpan priskripsi asal dan pulangkan Kad
Temujanji kepada pe-sakit untuk digunakan
Catat tarikh temujanji yang se-terusnya pada preskripsi asal dan
kembalikan kad temujanji dan pris-kripsi asal kepada pesakit
Ya
Tidak
Carta alir pemilihan pesakit untuk pembekalan ubat menggunakan Kad Temujanji
Carta alir proses pendispensan ubat untuk Kad Temujanji

PHARMACY BULLETIN VOL. 2/2014 DECEMBER 2014
Nicotine Patch Nicotine has been used as a smoking cessation aid for the relief of nic-
otine withdrawal symptoms and to ease craving1,2. It works by supplying ‘clean’
nicotine to the body. It contains around one-third to half the amount of nicotine
found in most cigarettes2. There are five types of NRT which are available and
approved by FDA, which are gum, nasal spray, inhaler, lozenges and patch2.
For nicotine patch, its nicotine delivery is
through transdermal system over either 16
or 24 hours, and the strength of nicotine
patch depends on individual body size and
smoking habits1,2,5.
Recommended starting dose5,6:
≥10 cigarettes a day and weight >45 kg: 21
mg/24 hour or 15 mg/16 hour patch
<10 cigarettes per day OR weight <45 kg
OR cardiovascular disease: 14 mg/24 or 10
mg/16 hour patch
Storage
Keep all the pouches in the box until immediately before use.
Store nicotine patch at temperatures below 25°C. Storage in a car in hot weather
may cause damage to the patch.
Keep nicotine patch out of reach of children.
Prepared by: Abdul Hakim Nordin Page 9
Counselling:
Side effects
Usually minor and transient, some
such as sleep disturbance,
dizziness, weight gain and
headache may be related to
stopping smoking
Application site reactions may
occur: redness, itch, rash – stop if
severe
Vivid dream (especially 24-hour
patch)

PHARMACY BULLETIN VOL. 2/2014 DECEMBER 2014
Counselling points1,3,4:
References
1. Australian Medicines Handbook 2014, Australian Medicines Handbook Pty Ltd; Adelaide.
2. Pahaki, NA 2013, Nicotine Replacement Therapy, Buletin Penawar, pg. 2, viewed 13 November 2014, < http://
hsajb.moh.gov.my/versibaru/uploads/farmasi/coverbuletin.pdf >
3. Pharmacia & Upjohn Pty Ltd, 1997, Nicorette Patch, brochure, Pharmacia & Upjohn Pty Ltd, Rydalmere.
4. Pharmaceutical Society of Australia (PSA) 2009, Fact Card Booklet 2009 (Pharmacy Self Care), ACT.
5. Sansom LN, ed, Australian pharmaceutical formulary and handbook, 22nd edition, Canberra: Pharmaceutical Society of
Australia, 2011.
6. QuitSA, n.d., Nicotine Replacement Therapy (NRT), brochure, QuitSA, SA.
Prepared by: Abdul Hakim Nordin Page 10

PHARMACY BULLETIN VOL. 2/2014 DECEMBER 2014
Prepared by: Abdul Hakim Nordin Page 11
AKTIVITI– aktiviti
KENALI UBAT ANDA 2014
13.02.2014
Penggunaan Kosmetik yang Bernotifikasi
Bilik Seminar Telepsikiatri, Hospital Permai
24.05.2014 Kenali Ubat Anda
Masjid Taman Anggerik, Kempas
11.06.2014
Kenali Ubat Anda (Ubat-ubat berdaftar)
Bilik Seminar Telepsikiatri, Hospital Permai
03.07.2014
Kenali Ubat Anda
Bilik Seminar CPU
12.10.2014
Kenali Ubat Anda (Bengkel Peningkatan Pesakit Ke Arah Penjagaan dan
Pemulihan di dalam Komuniti)
Bilik Seminar CPU
13.10.2014
Kenali Ubat Anda
Taman Sinar Harapan Jubli
16.10.2014
Kenali Ubat Anda
Auditorium, Kompleks Kementerian
Dalam Negeri, Setia Tropika
14.09.2014
Kenali Ubat Anda
Pertubuhan Kebajikan Insan
Istimewa Johor Bahru

PHARMACY BULLETIN VOL. 2/2014 DECEMBER 2014
28.10.2014
Pameran Secara Interaktif
Program Kesihatan Mental (Prokem) Bil 1/2014
Dewan Balai Polis Johor Bahru
11.06.2014
Pameran Kenali Ubat Anda
Foyer Kompleks Pesakit Luar Hospital Permai
16.10.2014
Pameran Kenali ubat Anda
Auditorium, Kompleks KDN, Setia Tropika
29.10.2014
Pameran Secara Interaktif
Program Kesihatan Mental (Prokem) Bil 2/2014
Foyer Perpustakaan UTM
23.11.2014
Pameran Secara Interaktif Program Kesihatan Mental
(Prokem) Bil 4/2014
Padang Kampung Pak Khalib, Pekan Nenas Pontian
4.11.2014
Pameran Secara Interaktif Program Kesihatan Mental
(Prokem) Bil 3/2014
Dewan Kolej Universiti Selatan
27.11.2014
Kenali Ubat Anda (Mental Health Karnival
anjuran Sapura Kenchana Holding
Mines, Selangor
25.04.2014
Pameran Kenali Ubat Anda
Ruang legar, Persada ICC
27.11.2014
Pameran Secara Interaktif Program Kesihatan
Mental (Prokem) Bil 5/2014
Ruang Legar Aras 3 HSIJB
AKTIVITI-aktiviti
KENALI UBAT ANDA 2014
Prepared by: Abdul Hakim Nordin Page 12

PHARMACY BULLETIN VOL. 2/2014 DECEMBER 2014
New drug in the formulary
T. Agomelatine 25mg
T. Sodium Valproate Chrono 500mg
T. Desvenlafaxine ER 50mg
Prepared by: Foo Qin Hao Page 13
X Q E P S U R Y C V I C V E D I G L P Y
R Z R I K T K O G V P G J L E M A A W U
R V U W N D Z I H C I R O O G O M F F V
P Z T G P D N E E D L E S E V O L G F C
K P C N H P P N L J L E H G R R K F A R
H R N G A V C X T B S D P Q D V V U J X
F Z I L R U O O Y G Y T D C N I E V X K
V P T R M N W H M N L A I P P Y O O F F
S A A E A B C O M P U T E R R R Y F A J
A P I G C O L O U S O S G A E O S D B I
V P E I I H A D U I L U S X S T P Z D H
A D I Y S J N B B A J N N I C N J Z Q Q
N B K G T I R I B D E J K D R E L E Z P
N Z T J K A T J C P O N J N I V W J R F
I A A O N N A Q S I K N I G P N A B I L
H Q L D A C O I Q B A Z U C T I G A P H
A Z Y S K O D I G G E N E R I C K R X Q
A Y S E L U S P A C E Q I S O D X J T F
U B T W I R X I M Y Y U Y X N N E N F T
Y X R O X P T O Y L L H F R B H N M K Y
PILLS TECHNICIAN PHARMACIST
MEDICINE TINCTURE
COMPOUNDING IVROOM
ANTIBIOTIC NEEDLES
CAPSULES COMPUTER
STAT DISPENSARY
PRESCRIPTION HOOD
GENERIC GLOVES BRAND
INVENTORY
Words List

![[XLS] · Web view1 2 3 10/20/2014 28 10/20/2014 32 10/20/2014 4 10/20/2014 20 10/20/2014 25 10/20/2014 29 10/20/2014 25 10/20/2014 21 10/20/2014 24 10/20/2014 18 10/20/2014 8 10/20/2014](https://static.fdokumen.site/doc/165x107/5ad5cadd7f8b9a0d2d8e19b7/xls-view1-2-3-10202014-28-10202014-32-10202014-4-10202014-20-10202014.jpg)